During prolonged operation of PEM water electrolyzer (PEMWE) in an acidic environment, the formation of TiO2 on the surface of the Ti-PTL causes passivation, resulting in an increase in the cell voltage.
Herein, the Ti-PTL treatment involved three steps: electrochemical etching to remove surface oxides, electrostriking to form an Au underlayer and electroplating to apply a protective top layer of Au. The Au was uniformly distributed on the top of the surface and inner structure of Ti-PTL under the optimized rotation speed of 50 rpm, denoted by Au/Ti felt [R = 50]. The Ti-PTL treatment under optimized rotation presented a more effective anticorrosive coating in simulated conditions of a PEMWE compared to treatments without rotation and those with high rotation of 200 rpm. In PEMWE cell, the Au/Ti felt [R = 50] PTL demonstrated a voltage reduction of 124 mV compared to Au/Ti felt [R = 0] at 2.0 A/cm2.
The corrosion study and durability measurements were conducted using a homemade simulated flow corrosion cell. 3D printing was employed to fabricate non-metallic bipolar plates (BPs) that included a simulated flow field structure at the PTL/BP interface. This approach paves the way to independently observe the impact on cell voltage caused by the degradation of the PTL.
1. Introduction
Hydrogen produced through electrolysis is a highly prospective energy carrier due to its high specific energy and its status as an environmentally sustainable fuel [1,2]. The Polymer Electrolyte Membrane Water Electrolyzer (PEMWE) is a device used to convert electrical energy into chemical energy by splitting water into oxygen and hydrogen. In a conventional PEMWE cell, a catalyst-coated membrane (CCM) is sandwiched between two porous transport layers (PTLs). These PTLs are supported by two metallic bipolar plates (BPs) and two electrically conductive current collectors [1,3,4]. Water undergoes splitting into oxygen, electrons, and protons at the anode. These protons migrate through the proton exchange membrane to the cathode, where they combine with electrons at the catalyst layer (CL) to generate hydrogen. The hydrogen-rich, low-potential reducing conditions minimize the corrosion of BPs and PTLs at the cathode. The thermo-neutral voltage of the PEMWE cell at ambient conditions is approximately 1.48 V on the anode side, while the operating voltage may exceed 2.00 V [5]. The CCM creates an intense acidic environment due to the fluorinated polymer with sulfonic acid side chains, especially at high current densities [[5], [6], [7], [8]]. Consequently, the metallic components, such as PTLs and BPs, will be susceptible to corrosion in an oxygen-rich anodic environment with a high applied potential and a low pH [2,7]. The PTL is more susceptible to degradation because it is positioned directly adjacent to the CCM [9]. The degradation of PTLs in an acidic environment can lead to increased interfacial contact resistance (ICR) and contamination of the CCM, and the CCM is a highly costly component of any PEMWE cell.
The PTL serves not only to transport water from the BPs to the CCM and gases from the CCM to the BPs, but also to facilitate the transfer of electrons and heat between the flow field structure of the BPs and the CCM. Therefore, an ideal PTL is anticipated to have promising anti-corrosive properties, negligible mass transport losses, and excellent electrical and thermal conductivity [1,10,11]. Titanium (Ti) fibers, known for their excellent corrosion resistance, are commonly used to fabricate PTL components in PEMWE cells. However, the passivation of Ti-based PTL occurs due to prolonged exposure to high applied anode potential. This leads to the oxidation and subsequent dissolution of most electron-conducting substrates, increasing the ohmic resistance of the device and raising the cell voltage [7]. To mitigate excessive passivation in Ti fibers, it is standard practice to use a protective coating of noble metals (such as Au, Pt, and Ir) [10,[12], [13], [14]]. This method improves electrical conductivity, enhances corrosion resistance, and reduces interfacial contact resistance [10,13,15].
Modifications in chemical-physical conditions, such as variations in protective layer thickness, oxidation state, degree of corrosion, and dissolution, are particularly important because of their influence on electrical conductivity and their implications for durability [16]. The advancements in coating methods are engineered by identifying these correlations, enabling cost-effective materials and components to achieve optimal operation of the cells. Rakousky et al. resolved these challenges through the deposition of a platinum layer onto a sintered porous Ti felt, effectively applying it as a corrosion-resistant layer to inhibit oxidation [12]. A sputtered Ir layer was applied onto a Ti felt by Liu et al. to control the Ti fiber oxidation [10]. Srour et al. explored the effects of PVD-deposited noble metal coatings, including Pt, Au, and Ir, and concluded that Pt coatings provided excellent beginning-of-life performance, while Ir coatings demonstrated superior durability during start-stop events [17]. Using Pt and Ir coatings increases the capital costs of PEMWE cells, and the coating quality depends significantly on sputtering parameters. Coating high-aspect-ratio surfaces may result in some areas remaining uncoated due to the shadowing effect [18].
Sputtering requires costly vacuum systems, specialized chambers, and substantial operational and maintenance costs. The setup and maintenance of such equipment can be expensive, making it less economically viable for large-scale or low-cost production. Furthermore, during the sputtering process, a significant portion of the precious metal target material is lost as waste rather than being deposited onto the substrate. This leads to lower material efficiency and higher costs, particularly when working with expensive metals such as Ir, Pt, and Au. The electroplating process is a practical approach to depositing a gold layer as a protective film on the PEMWE cell components [15,19]. This method is well-established and relatively straightforward, leading to lower processing costs than Pt and Ir. The electroplating of gold is a mature technology and easy to implement for large-scale coating [20,21]. The electroplating process is affected by several factors, such as the surface property (hydrophilic/hydrophobic) of the substrate, ingredient of plating solution, arrangement of electrodes, current distribution, and operating parameters [20]. In the present study, convection was induced by the rotation of the substrate during electroplating, thereby facilitating uniform gold deposition throughout the inner structure of the porous Ti felt.
Typically, when the PEMWE cell was disassembled after a prolonged demonstration, it was noted that fragments of the protective layer had detached and adhered to the anodic catalyst layer of the CCM [5,7]. Detachment could be attributed to corrosion processes occurring at the contact area, which diminish adhesion between the substrate and protective layer. Cherevko et al. have studied the extent of noble metal (Pt, Au) corrosion in acidic media [22]. They observed that corrosion of noble metal and the oxygen evolution reaction (OER) are related, and the rate of both processes increases significantly under high applied potential [23].
Beyond observing the coating textural property on the surface of the PTL fibers, it is pertinent to investigate the anticorrosion property and lifetime of the protective layer. Conventional laboratory-scale corrosion studies, which employ stagnant solution and three-electrode assembly, may not offer precise information due to the omission of critical parameters related to actual cell-operating conditions or the arrangement of PEMWE cell components [19,24,25]. The PTL is fixed just adjacent to CCM, where oxygen evolution occurs. Hence, water flow plays a significant role in the degradation of coated PTL. Furthermore, designing BPs with proper flow field structure is necessary to provide simulated flow conditions across the PTL. During PEMWE operation, metallic PTL, and metallic BPs are susceptible to corrosion, making it difficult to differentiate the corrosion of individual components. Additive manufacturing (3D printing) enabled rapid and reproducible prototyping of the non-metallic electrolyzer components, improving cost factors and manufacturability [[26], [27], [28]].
Herein, the gold electroplating on the surface of porous Ti felt was performed under different convective conditions. The substrate was rotated during the electroplating of Ti-PTL to create convective media across the porous Ti felt. The corrosion study and measurement of the durability of the gold coating were performed using a homemade flow corrosion cell. In this work, a 3D printing technique is used to fabricate non-metallic bipolar plates consisting of flow field channels. The 3D printed channel provided the simulated flow field structure on the interface of PTL and BPs. The simulated conditions, such as the concentration of the solution (0.5 M H2SO4), temperature (80 °C), the flow rate of the solution (50 mL min-1), the serpentine flow field structure of BPs, and operating cell potential (2.0 V) were considered as in any conventional PEMWE. The gold-coated PTL was also examined in situ by polarization and electrochemical impedance spectroscopy (EIS) techniques using a conventional PEMWE cell. The results indicate that gold electroplating under convective conditions is a simple and reliable strategy for treating Ti felt and enhancing the PEMWE cell performance.
1.1. Materials and methods
The Ti felt of thickness 250 μm (Bekaert, Grade I titanium, 68% porosity, mesh openings up to 100 μm, fiber thickness 25 μm) was employed as PTLs for gold coating and subsequent testing in PEMWE cell. The surface passivating TiOx layer forms naturally through exposure to ambient air during fabrication and handling. As shown in Fig. 1 (a), First, the compact TiOx passivation layer is removed by electrochemical etching process, which facilitated a strong adhesion of the protective gold coating on the Ti felt. Second, electrostriking was performed to deposit the thin gold underlayer. Third, electroplating was performed to deposit a thick gold protective layer.
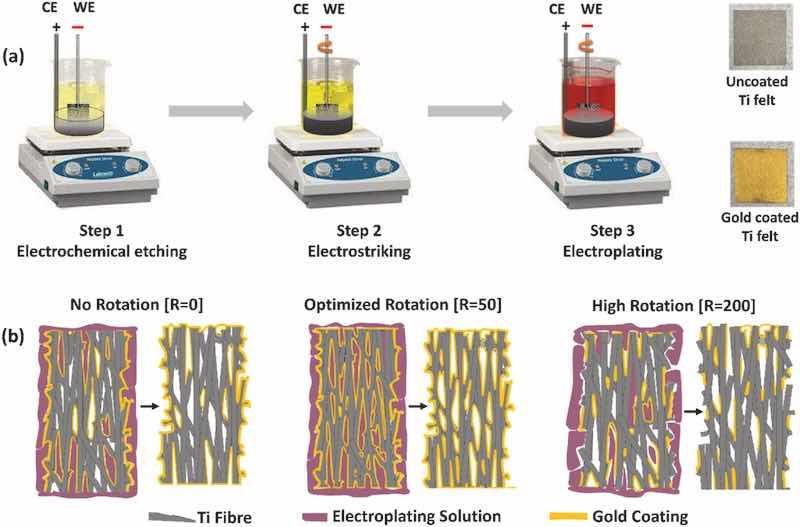
Fig. 1. (a) Schematic diagram of gold coating on the Ti felt. (b) Schematic diagram of effect of substrate rotation during electroplating. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
The cylindrical design of the counter electrode facilitated the uniform current distribution during the treatment of Ti felt (In electrochemical etching, electrostriking, and electroplating process). The electrochemical etching, electrostriking, and electroplating were carried out using the electrochemical workstation (Biologic, SP 150). In the electrochemical etching step, Ti felt is used as the cathode (working electrode), and cylindrical platinized Ti mesh (Fuel Cell Store) is used as the anode (counter electrode). An acidic solution of 0.5 M H2SO4 is maintained at 60 °C, and a negative cathodic current density of 0.1 A/cm2 was applied for 10 min and 20 min. In the electrostriking and electroplating process, the working electrode (Ti felt) is rotated to increase the electrolyte exposure of the inner structure of porous Ti felt. Additionally, the rotation of the Ti-felt substrate also facilitated the detachment of hydrogen bubbles during the electroplating process. The rotation effect was studied at three speeds of 0, 50, and 200 rpm, as shown in Fig. 1 (b). In electrostriking process, the working electrode (etched Ti felt) and cylindrical counter electrode (platinized Ti mesh) are immersed in 24K acid gold strike solution (TriVal, TV128, Gold Plating Services). The gold strike solution was maintained at 22 °C, and a negative voltage of 5 V was applied for 30 s. In the solution, where the pH was below 1, the active species present was potassium tetracyanoaurate (KAu(CN)4), which allowed the electrochemical reduction of gold on the electrochemical etched Ti felt. A thin, faintly yellow-tinted gold layer was uniformly deposited and adhered well to the titanium felt surface. This electrostriked gold layer provided the electrically conductive sites for efficient deposition of a protective gold coating during electroplating.
In electroplating process, the working electrode (gold strike Ti felt) and cylindrical counter electrode (platinized Ti mesh) were immersed in 24K bright gold solution (Gold Plating Services). The bright gold solution is maintained at 40 °C, and a negative voltage of 3 V was applied for 300 s. During the electroplating step, the pH was between 4 and 5, and the potassium dicyanoaurate (KAu(CN)2) served as the active component, which offered the uniform gold deposition on the inner and outer structure of the porous Ti felt. In each step treated Ti felt was thoroughly rinsed using DI water. The sample prepared with no rotation of substrate (0 rpm), with rotation of 50 rpm, and 200 rpm are denoted as Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200], respectively.
1.2. Physical characterization
The surface morphology and elemental composition of the uncoated Ti felt, and gold-coated Ti felts were studied by scanning electron microscopy (SEM; Hitachi-Suisio, acceleration voltage 20 kV, working distance 15 mm, and emission current 100 μA) and energy-dispersive X-ray spectroscopy (EDS). The cross-sectional analysis of the PTLs was performed to measure the thickness of the gold coating on the Ti felt. The cross-section of the sample was prepared using a focused ion beam (FIB) in a ThermoFisher Helios 5 UXe Dual Beam Plasma FIB-SEM. The crystallinity and phase analysis were carried out by X-ray diffraction (XRD) on a Bruker D8 Focus X-ray diffractometer with Cu Kα (λ = 0.15418 nm) radiation source. The gold loading on the Ti felt was measured using X-Ray fluorescence (XRF, Niton XL2 XRF Analyzer). The X-ray photoelectron spectroscopy (XPS) was conducted to examine the surface chemical composition of the gold-coated PTLs, focusing particularly on the properties of the gold in the tested samples. The XPS data was recorded using a Thermo-VG Scientific ESCALab 250 microprobe (150 W Al Ka radiation source).
To characterize the electrical conductivity properties of the uncoated and gold-coated Ti felt (PTL), interfacial contact resistance (ICR) measurements were conducted using varying compression pressures. The ICR was measured using a compression setup (Zwickroel, Germany), in which the samples were compressed between two copper plates coated with gold. The compression range was applied from 0.5 to 5 MPa, and measurements were conducted at room temperature. The combined resistance associated with PTL and the two contact resistances between the PTL and gold-plated copper sheets were calculated by applying a DC of 4 A in conjunction with the evaluated potential and Ohm's law.
1.3. Simulated durability test of gold-coated Ti felt
The simulated durability test (SDT) was employed to assess the lifespan of the gold coating on Ti felt. The test was conducted under the simulated environment of a PEMWE cell, and harsher operating conditions were imposed to accelerate the coating degradation process. The SDT of gold-coated PTL was conducted in a two-electrode assembly using a DC power supply (BK Precision, 9115 series). The gold electroplated Ti felt was assembled as the anode, and a platinum mesh as the cathode. As shown in Fig. 2 (a, b) and Fig. S1, the anode and cathode were fixed adjacent to 3D printed flow field channel, which provided the simulated flow field structure on the interface of PTLs and BPs. The working area of both anode and cathode was 5 cm2. The acidic solution was transported to/from the flow cell by a peristaltic pump (Precision peristaltic pump, L100-1S-2) with a constant flow rate of 50 mL min−1 through Tygon tubing (3.8 mm inner diameter) from a reservoir of 0.5 M H2SO4 to the flow field channels of the BPs. The simulated durability test was carried out at 1.0 A/cm2, 80 °C, with a 2 cm distance between the anode and cathode. At a current density of 1 A/cm2, both the acidic solution and oxygen are present at the anode side, simulating real operating conditions of the PEMWE cell. The 3D-printed perforated separator was fixed between the anode and cathode to reduce the crossover of oxygen and hydrogen gas bubbles. The sharply increased voltage with respect to time indicates the durability of the coating.
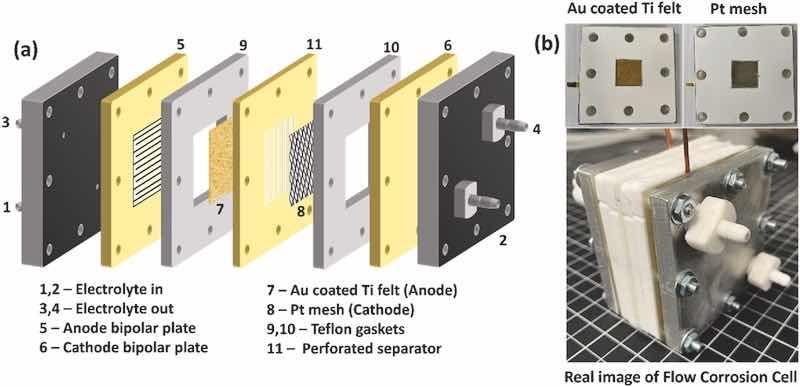
Fig. 2. (a) Schematic diagram of the simulated durability test device, (b) real image of simulated flow electrochemical device.
1.4. Corrosion test evaluation of uncoated and gold-coated PTL
The corrosion tests were performed using above mentioned flow electrochemical setup controlled with an electrochemical workstation (Biologic, SP 150). Stabilization under open circuit voltage (OCV) is a reference for the electrochemical behavior of the material, particularly when studying corrosion or passivation properties. Subsequently, potentiodynamic tests were conducted to investigate the corrosion properties of uncoated Ti felt and gold-coated Ti felt using potential scans up to 2.0 V. The selected voltage window enabled the evaluation of corrosion experienced by PTLs on the actual anode side of a PEMWE cell. The potentiostatic test was performed for 10 h at 2.0 V. Typically, these tests are conducted at voltages close to the OCV. However, in this case, experiments were designed to assess the behaviour of the coated PTLs at the typical operating potential of the PEMWE cell, which is around 1.7–2.2 V. The EIS analysis was carried out at OCV across the frequency range of 100 kHz to 0.1 Hz with a perturbation amplitude of 10 mV. After the potentiostatic test at 2.0 V, the gold dissolution was examined using inductive coupled plasma – atomic emission spectrometry (ICP-AES, Agilent 8900).
1.5. PEM water electrolysis test
The performance of gold coating on Ti felt was evaluated in-situ by recording polarization curve using PEM electrolyzer (Scribner Electrolyzer, Cell-5). Deionized water (Milli-Q water, 18.2 MΩ cm) was transported to the anode side of the PEMWE cell at a flow rate of 50 mL min−1. A commercial CCM was purchased from Ion Power Company, and according to the specification sheets, it includes Nafion 115 as the membrane. The IrOx loading at the anode is 1.0 mg cm⁻2, and the Pt/C loading at the cathode is 0.5 mg cm⁻2. The working area of the CCM is 5 cm2. Uncoated and gold-coated Ti felts were assembled as the PTL at the anode of a PEMWE cell. In all experiments, carbon fiber paper with MPL (Fuel cell store, AvCarb GDS3250, nominal thickness 225 μm) was assembled as the cathodic PTL. Platinum-coated BP with single serpentine flow fields was employed on the anode side, and graphite plate with single serpentine flow fields was employed on the cathode side. The PEMWE cell was tightened using 250 μm thick PTFE gaskets on both the anode and cathode, with a torque of 8 N m applied to each of the eight bolts. The PEMWE cell was connected to the Greenlight electrolyzer test station (E30) to record the polarization curve and EIS spectrum. Once the temperature of 80 °C was reached, the conditioning of the cell was performed by applying each current density of 0.2 A cm−2, 0.4 A cm−2, 0.6 A cm−2, 0.8 A cm−2, and 1 A cm−2 for 30 min. Afterward, the j-V test was recorded by monitoring the potential as the current density incrementally increased from 0 to 2 A/cm2. The EIS was performed at a current density of 1 A/cm2 in the frequency range of 100 kHz to 0.1 Hz with an AC amplitude of 0.2 A/cm2. The corrosion study, simulated durability analysis, and performance analysis were conducted using the freshly prepared PTL.
2. Results and discussion
2.1. Surface morphology and elemental composition study of gold-coated Ti felt
The surface morphologies of the gold-coated Ti felts were studied using SEM. As shown in Fig. 3(a) and the inset, the uncoated Ti fibers are smooth before treatment. The formation of a titanium hydride (TiHx) layer was observed on the uncoated Ti-felt surface after the electrochemical etching in acidic media. In Fig. S2 (a), the XRD spectra of electrochemically etched samples showed diffraction peaks at 35.8°, 41.1°, and 59.3°, which corresponds to the presence of TiHx on the surface of Ti felt [[29], [30], [31]]. The electrochemical etching of the porous Ti felt decreased the contact angle of the uncoated Ti felt. The electrochemically etched sample (10 min) presented a decrease in contact angle (28.3°) as compared to the electrochemically etched sample (59.1°), as shown in Fig. S2 (b). The presence of TiHx on the surface exhibited enhanced wettability on the Ti substrate [32,33]. The increased wettability of the uncoated Ti felt facilitated the efficient electrochemical active sites on its inner and outer structures during the subsequent electrostriking and electroplating steps. The electrochemical etching of the Ti felt for a longer duration of 20 min, further presented an increase in contact angle (38.1°). The cathodic polarization of Ti substrate for a long time may cause hydrogen embrittlement, which may create cracks on the surface and exposure of uncoated Ti felt [34,35].
![Fig. 3. SEM images of fresh (a) uncoated Ti felt, (b) Au/Ti felt [R = 0], (c) Au/Ti felt [R = 50], and (d) Au/Ti felt [R = 200] samples, inset shows magnified view of respective SEM image.](/images/images/whitepapers/ptl/3.jpg)
Fig. 3. SEM images of fresh (a) uncoated Ti felt, (b) Au/Ti felt [R = 0], (c) Au/Ti felt [R = 50], and (d) Au/Ti felt [R = 200] samples, inset shows magnified view of respective SEM image.
Fig. 3 (b, c, and d) shows the SEM images of the gold-coated Ti felt under rotation speeds of 0, 50, and 200 rpm. The gold is uniformly distributed on the top of the surface and inner structure of porous Ti felt under the optimize rotation speed of 50 rpm. However, some pin holes formed throughout the top of the surface, and an uncoated area on the inner structure of the Ti felt under the without rotation condition (0 rpm). The high rotation speed (200 rpm) was also not favorable to depositing anticorrosive protective film with strong adherence. The high rotation speed facilitated the gold deposition on the inner structure of Ti felt but impeded the formation of compact gold film on the top of the surface. It can be found that under an optimized rotation speed of 50 rpm, the gold is distributed uniformly on both the outer boundaries and the inner volume of the porous Ti fiber. The thickness of the gold coating on the Ti felt was approximately 180 nm, 150 nm, and 63 nm for the Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, respectively (Fig. S3). The optimized rotation during the Ti-PTL treatment resulted in a gold loading of 0.2 mg cm⁻2, while the samples treated with no rotation and high rotation speed showed loadings of 0.36 mg cm⁻2 and 0.11 mg cm⁻2, respectively.
The EDS line scanning was performed to analyze the gold coating on porous Ti felt's inner and outer structure. The high peak intensity of Ti in Fig. 4 (a, c) is attributed to the uncoated area and pin holes formed on the surface of gold-coated Ti felt. The Ti peak intensity is small in Fig. 4 (b), which indicates that the compact gold layer is distributed more uniformly throughout the surface under optimized rotation conditions. The EDS line scanning confirmed that the Ti-PTL treatment without substrate rotation was not facilitating the gold coating on the inner structure of porous Ti felt, as shown in Fig. 4 (a). In addition, as shown in Fig. 4 (c), the high rotation of the substrate during Ti-PTL treatment did not provide a compact uniform gold coating on the top of the Ti fibers. Furthermore, in Fig. 4 (b), the EDS line scanning showed that the Ti-PTL treatment under optimized rotation showed the uniform gold coating on inner and outer structure of porous Ti felt.
![Fig. 4. EDS line scanning spectrum of fresh (a) Au/Ti felt [R = 0], (b) Au/Ti felt [R = 50], and (c) Au/Ti felt [R = 200] samples.](/images/images/whitepapers/ptl/4.jpg)
Fig. 4. EDS line scanning spectrum of fresh (a) Au/Ti felt [R = 0], (b) Au/Ti felt [R = 50], and (c) Au/Ti felt [R = 200] samples.
2.2. Corrosion analysis of gold-coated PTL
In addition to the conventional examination of the coating properties on the surface of the Ti fibers, it is essential to examine the interface between the Ti fiber and the anti-corrosive gold layer. Investigating this interface under simulated PEM electrolyzer conditions will provide insights into the interrelated mechanisms affecting various materials and components within the cell. The precise information for a PEM water electrolyzer may not be obtained through traditional lab-scale electrochemical analysis, which uses stagnant solution and three-electrode cell configurations. The three-electrode setup does not account for critical parameters relevant to the actual operating conditions of the cell or the contributions of other key components, including the BPs, membrane, catalyst layer, and ionomer, to the PTL. Consequently, it is imperative to connect the data obtained from laboratory-scale testing with the long-term performance of real PEMWE cells through engineered characterization techniques.
In this work, the anode and cathode were fixed adjacent to a non-electrically conductive 3D printed flow field channel, which provided the simulated flow field structure on the interface of PTL and BPs. The 3D printed flow channels mimicked the water transport and oxygen removal through the porous Ti felt as present in conventional PEM water electrolyzer [19,25]. The simulated conditions such as solution temperature (80 °C), the flow rate of the solution (50 mL min−1), the concentration of the liquid (0.5 M H2SO4), serpentine flow field structure of BPs, and operating cell potential (2.0 V) were considered as in any conventional PEM electrolyzer [10,15,36].
The electrochemical corrosion studies were designed to investigate the PTL behaviour in the anodic simulated environment of a PEM water electrolyzer. Potentiodynamic studies were conducted in a flow corrosion cell, and the results were compared with the uncoated Ti felt. As shown in Fig. 5 (a), the corrosion current density of the Au/Ti felt [R = 50] (0.083 μA/cm2) is 30 times, 5 times, and 2 times lower as compared to uncoated Ti felt (2.5 μA/cm2), Au/Ti felt [R = 200] (0.41 μA/cm2), and Au/Ti felt [R = 0] (0.17 μA/cm2), respectively. The large corrosion current density of the Au/Ti felt [R = 0] and Au/Ti felt [R = 200] sample was due to the penetration of acidic solution to the Ti substrate through the pinholes and cracks in the gold coating. The uncoated Ti felt showed a corrosion potential of −0.75 V, followed by continuous passivation from the oxide layer. In contrast, the Au-coated layer has an extraordinary impact on the corrosion performance of the PTLs. The Au/Ti felt [R = 50] sample presented a corrosion potential of −0.37 V, significantly more significant than the uncoated Ti felt. The results depict that the Au coating offered an excellent anti-corrosive property with high potential in potentiodynamic tests. In contrast, a higher anodic current value between −0.75 V and 2.0 V in the uncoated Ti felt was noted, which is the lowest observed value between −0.37 V and 2.0 V in the Au/Ti felt [R = 50] sample. This improvement may be attributed to the enhanced stability of the passive film and the increased re-passivation property of the Ti substrate achieved through gold electroplating, compared to the uncoated Ti substrate, as other authors have also indicated [15,19,37].
![Fig. 5. Corrosion analysis of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, (a) Tafel potentiodyanamic polarization curve, (b) EIS spectra, (c) potentiostatic polarization curve at 2.0 V, and (d) OCV measurement.](/images/images/whitepapers/ptl/5.jpg)
Fig. 5. Corrosion analysis of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, (a) Tafel potentiodyanamic polarization curve, (b) EIS spectra, (c) potentiostatic polarization curve at 2.0 V, and (d) OCV measurement.
The corrosion resistance was analyzed using the Nyquist and Bode plots, as shown in Fig. 5 (b) and Fig. S4, respectively. The inset in Fig. 5 (b) presents the equivalent circuits that fit the EIS data from uncoated Ti felt and gold-coated Ti felt samples. Rs depicts the solution resistance, Cdl stands for double-layer capacitance, and Rct is the charge transfer resistance between the Ti felt and corrosion media. The CPE presents the constant phase element due to inhomogeneity in the gold coating. Rgc presents an additional corrosion resistance due to the gold coating on the porous Ti felt. The Nyquist plots showed a larger diameter of the semicircle in the case of gold-coated Ti felt as compared to uncoated Ti felt. It indicates that an additional corrosion resistance (Rgc) facilitated a high charge transfer resistance between the Ti felt and corrosion media in simulated PEM electrolyzer conditions.
In Fig. S4, the phase angle plot of the gold-coated Ti felt in the high-frequency region was merged with uncoated Ti felt due to the presence of local defects on the surface, but in the low-frequency region, the phase angle of the gold-coated Ti felt was larger as compared to uncoated Ti felt. The large phase angle value at the low-frequency region depicts the high capacitive performance [24,38]. In the case of the Au/Ti felt [R = 50] sample, the gold coating was more capacitive and led to lower penetration of the acidic solution as compared to uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] samples. Table S1 presents the Rs, Rct, and Rgc value obtained from the equivalent circuit. The value of Rs is lowest in the case of the Au/Ti felt [R = 50] sample, which is due to the crack-free coating of gold, owing to efficient electron transfer from the anode surface to the current collector. The value of Rct in the case of Au/Ti felt [R = 50] samples was higher as compared to uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] samples. It indicates that the Ti-PTL treatment under optimized rotation of Ti felt substrate provided an excellent anti-corrosive gold layer. The highest value of Rgc in the Au/Ti felt [R = 50] sample indicates that gold electroplating can increase the protective film's corrosion resistance in the anode of the PEM water electrolyzer. In the corrosion study, the data obtained from the Nyquist plot and the Bode plot support the results from the potentiodynamic polarization study.
To further evaluate the feasibility of gold electroplating on the porous Ti felt, chronoamperometry measurements at 2.0 V were performed to examine the stability of Au-coated Ti felt samples in a simulated anodic condition for 10 h, as shown in Fig. 5 (c). Based on the potentiostatic polarization curves, the current density of uncoated Ti felt decreases rapidly within the first 15 min and stabilizes at a constant level. This abrupt change in current density may be caused by electrochemical cleaning of the Ti surface and erosion of the oxide layer due to the oxygen bubbles generated. Afterward, the oxide TiOx starts to nucleate due to the anodization of the base Ti surface at high potentials. In addition, the exponential decrease might be due to the oxidation of the Ti surface or the dissolution of the oxide layer in the acidic solution, which resulted in the formation of the protective oxide layer. In the case of the best optimized gold-coated sample (Au/Ti felt [R = 50]), the current density under potentiostatic polarization reduced sharply, and it showed the lowest steady-state current. The results reveal that the Au-coated Ti felt shows good stability. Therefore, forming a stable Au-coating layer can notably boost the anti-corrosive property.
In Fig. 5 (d), the uncoated Ti felt, Au/Ti felt [R = 200], Au/Ti felt [R = 0], and Au/Ti felt [R = 50] showed open circuit voltage (OCV) of −0.8 V, −0.6 V, −0.35 V, and −0.2 V, respectively. The gold-coated sample Au/Ti felt [R = 50] showed more positive OCV than uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] samples. The positive shift of OCV is typically attributed to the anti-corrosive property of the gold layer on the porous Ti felt [39]. This indicates that a gold layer has a better passivation possibility in an acidic solution due to losing a small number of sacrificial electrons under OCV conditions [40,41]. Furthermore, the OCV shifted to more positive potential in time after the potentiostatic test for 10 h at 2.0 V. After polarization at 2.0 V, the OCV shifted to more positive values for both uncoated and gold-coated Ti felt. The positive shift in OCV was due to the transition of metallic Ti0 to the Ti4+-oxides passivation layer [42]. The formation of the Ti4+-oxides passivation layer was also confirmed by XPS analysis (section 3.3). Although the formed passivation layer may act as a protective layer, the Ti4+−oxide layer has limited electrical conductivity, which may impede the performance of any PEM water electrolyzer [[43], [44], [45]]. After corrosion testing, the maximum exposed uncoated area with an acidic solution showed a high positive value of OCV. The highest positive OCV value of 0.7 V was obtained for uncoated Ti felt. After that, Au/Ti felt [R = 200] showed an OCV value of 0.6 V due to significant corrosion and dissolution of gold coating from porous Ti felt in an acidic solution.
The corrosion analysis alone is insufficient to prove that gold coating protects the Ti felt against corrosion. In literature, there are many studies where the dissolution of noble metals (such as Pt, Ir) from CCM is studied by inductively coupled plasma–atomic emission spectrometry (ICP-AES) [7,46,47]. If there is indeed any release of Au and Ti ions from the gold-coated Ti felt, it should be present in the acidic solution. The ICP-AES was performed to investigate the Au and Ti dissolution in the acidic solution sample. As shown in Table 1, the gold dissolution of the Au/Ti felt [R = 0] sample was high at a constant potential of 2.0 V. In contrast, the Au/Ti felt [R = 50] showed the lowest quantity of Au in the acidic solution, confirming the absence of impactful corrosion of the Ti felt during the test. In the case of the Au/Ti felt [R = 200] sample, Ti was found in a higher concentration as compared to Au/Ti felt [R = 0] and Au/Ti felt [R = 50]. The uncoated Ti felt showed the highest concentration of Ti. This element source was porous Ti felt, where the uncoated area was directly contacted with an acidic solution.
Table 1. The quantity of Au and Ti in the acidic solution after potentiostatic test.
| Sample | Au (mg/L) | Ti (mg/L) |
| Au/Ti felt [R = 0] | 0.19 | 1.30 |
| Au/Ti felt [R = 50] | 0.068 | 0.081 |
| Au/Ti felt [R = 200] | 0.098 | 3.12 |
| Uncoated Ti felt | – | 5.43 |
2.3. Ex-situ testing of gold coated PTL after corrosion analysis
Each gold coated Ti felt is tested in a flow electrochemical corrosion cell. After the potentiostatic test electrochemical cell was disassembled and the surface of the gold coated Ti felts are examined ex-situ by SEM, EDS, XRD, and XPS. The gold coated Au/Ti felt [R = 50] PTL sample showed stable and lower corrosion current density up to 10 h as compared to uncoated Ti felt, Au/Ti felt [R = 200] and Au/Ti felt [R = 0] PTL samples. The stable performance was owed to uniform and strong adhesive gold coating on the porous Ti felt. Fig. 6 (b) shows a post operation SEM image of the Au/Ti felt [R = 50] PTL sample, there is negligible change in morphology even after potentiostatic testing of 10 h. On the other hand, as shown in Fig. 6 (a), in the case of the Au/Ti felt [R = 0] sample, the uncoated Ti fiber surface was clearly visible due to the crystallization of the gold layer and its segregation into interconnected islands on the coated gold crystals [22,23]. The dissolution of the protective layer was attributed to the disintegration of the coating in the acidic solution. In addition, as shown in Fig. 6 (c), in case of Au/Ti felt [R = 200] sample, there are many sites where gold was not coated, which resulted to pitted structures that emerged to be Ti-oxides after the potentiostatic measurement. This suggests that the surface of the Ti metal on the uncoated portions of the Ti fibers experienced oxidation during the potentiostatic test. As shown in Fig. S5, after the potentiostatic test the uncoated PTL exhibited the formation of a TiOx layer on the surface of the Ti felt.
![Fig. 6. SEM images of potentiostatic tested samples at 2.0 V for 10 h (a) Au/Ti felt [R = 0], (b) Au/Ti felt [R = 50], and (c) Au/Ti felt [R = 200], (d) XRD spectra of fresh and tested gold coated Ti felt samples. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)](/images/images/whitepapers/ptl/6.jpg)
Fig. 6. SEM images of potentiostatic tested samples at 2.0 V for 10 h (a) Au/Ti felt [R = 0], (b) Au/Ti felt [R = 50], and (c) Au/Ti felt [R = 200], (d) XRD spectra of fresh and tested gold coated Ti felt samples. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
The XRD spectra of the uncoated Ti felt and gold-coated Ti felt samples are shown in Fig. 6 (d). The XRD analysis of uncoated Ti felt revealed principal peaks at 35.1°, 36.9°, 40.1°, 53°, 71°, 76.1°, and 77°, corresponding to the (100), (002), (101), (102), (103), (202), and (201) planes, respectively. The diffraction peaks were well-fitted with the result of commercially pure titanium [43]. The gold-coated Ti felts showed additional peaks corresponding to (111), (200), (220), and (311) at 2θ angles of 38.1°, 44.3°, 64.8°, and 77.6°. The observed diffraction peaks of gold crystal structure assigned to JCPDS file number [04–0784] [48].
After disassembling the flow corrosion cell, it was analyzed that small portions of the Au layer had delaminated. The detached gold was mixed in a 0.5 M H₂SO₄ solution, which was confirmed by ICP-AES analysis. This effect was similarly observed in the case of the noble metal corrosion [22,23]. As shown in Fig. 6 (d), after the corrosion test, the diffraction peaks of gold remained only for Au/Ti felt [R = 50]. The gold layer with pin holes, cracks, and non-uniformity was susceptible to electrochemical corrosion. The detachment of the gold layer could be increased by corrosion processes at the non-uniform contact area, reducing the adhesion between the base substrate and the gold layer [13]. The detachment of the Au anticorrosion layer resulted in both increased ohmic resistance and mass transfer resistance in the PEM water electrolyzer [5,8]. At the locations where the Ti felt had detached its Au coatings, corrosion protection was compromised, likely resulting in increased contact resistance due to the passivation of the Ti fibers.
The XPS measurement was carried out to study the change in the oxidation state of metallic components (Ti and Au) before and after potentiostatic testing. The XPS profile of fresh uncoated Ti felt in Fig. 7 (a) shows a mixed metallic Ti0 and passivation TiO2 layer. The TiO2 surface-passivation layer was naturally formed through ambient air exposure during the fabrication and handling step [3]. After the corrosion testing, the Ti0 metallic peaks (Ti0 2p1/2 at 459.7 eV and Ti0 2p3/2 at 453.7 eV) were diminished, while only the oxide Ti4+ peaks (Ti4+ 2p1/2 at 465.1 eV and Ti4+ 2p3/2 at 459.2 eV) was dominant, Fig. 7 (c) [49,50]. This indicates that corrosion testing under harsh acidic conditions promotes the oxidation of the base Ti substrate to the TiO2 layer at the surface. In Fig. 7 (b), gold-coated Ti felt showed metallic Au0, indicating Au0 4f5/2 at 87.6 eV and Au0 4f7/2 at 83.8 eV [51]. After the corrosion testing, mixed metallic Au0 and Au3+ oxides were observed, indicating Au3+ 4f5/2 at 89.4 eV and Au3+ 4f7/2 at 85.8 eV, Fig. 7 (d) [52,53].
![Fig. 7. XPS spectra of Au/Ti felt [R = 50] sample, (a) fresh Ti 2p (b) fresh Au 4f, (c) tested Ti 2p, and (d) tested Au 4f.](/images/images/whitepapers/ptl/7.jpg)
Fig. 7. XPS spectra of Au/Ti felt [R = 50] sample, (a) fresh Ti 2p (b) fresh Au 4f, (c) tested Ti 2p, and (d) tested Au 4f.
The gradual increase in the dissolution of gold and the formation of a monolayer of gold oxide (Au3+) are linked together [54]. The high applied potential of 2.0 V in acidic conditions favored the formation of monolayer gold oxide, specifically under OER conditions [23]. The XPS spectra of the Ti and Au after the corrosion testing present a dominant presence of Ti and Au oxides compared to pristine gold-coated Ti felt. The diffraction peaks of Au oxide were not observed through XRD due to the growth of a thin layer of amorphous surface oxides. Since most of the bulk gold remains in a metallic state, the gold coating ensures optimal conditions for maintaining standard electrical conductivity and anti-corrosive properties [19,22]. This indicates that the gold layer underwent an oxidation process, likely occurring during the initial stage of operation due to oxygen evolution under harsh acidic conditions. However, further oxidation of the metallic gold was not extended into the bulk during the potentiostatic study [23,54].
2.4. Simulated durability test of gold-coated samples
The simulated durability tests (SDT) were performed using the flow corrosion cell controlled with a DC source meter. The simulated durability tests were performed under the same operating conditions (temperature, solution concentration, and flow rates) as the corrosion tests. The PTL on the anode side is exposed to harsh oxidizing conditions due to the high cell voltage and oxygen gas generation environment in conventional PEMWE cells [1,8]. In this work, the SDT was conducted to estimate the lifetime of gold coating on the Ti felt. The lifetime of the coating was calculated at an operating current density of 1.0 A cm−2. The sharply increased voltage indicates the durability of the coating under simulated conditions. The failure of the coating is primarily due to the gradual loss of the protective layer and the corrosion and degradation of the porous Ti felt. These factors contribute to the increase in potential observed during the simulated durability test. The uncoated Ti felt exhibited a rapid increase in voltage at 1 A/cm2, with the highest voltage reaching within 10–15 s (Fig. S6). As shown in Fig. 8 (a), the simulated durability of Au/Ti felt [R = 50] is 4 times to Au/Ti felt [R = 200] and 2 times to Au/Ti felt [R = 0]. In the case of Au/Ti felt [R = 50], there are a small number of uncoated nucleation sites for corrosion of base Ti felt substrate under harsh acidic conditions. The optimized rotation of Ti felt (R = 50 rpm) during Ti-PTL treatment improved the bonding energy between the gold coating and the porous Ti felt. Therefore, the durability of the protective layer was extended.
![Fig. 8. (a) Simulated durability test of Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, (b) ICR measurement of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, both before and after potentiostatic test, (c) Polarization curve, (d) Tafel slope analysis, (e) EIS, and (f) high frequency resistance plots of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] PTLs in PEMWE cell.](/images/images/whitepapers/ptl/8.jpg)
Fig. 8. (a) Simulated durability test of Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, (b) ICR measurement of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] samples, both before and after potentiostatic test, (c) Polarization curve, (d) Tafel slope analysis, (e) EIS, and (f) high frequency resistance plots of uncoated Ti felt, Au/Ti felt [R = 0], Au/Ti felt [R = 50], and Au/Ti felt [R = 200] PTLs in PEMWE cell.
Fig. 8 (b) illustrates the ICR of gold-coated Ti felt before and after the potentiostatic test. The fresh uncoated Ti felt showed a high contact resistance of 9.5 mΩ cm2 at 1.5 MPa. In the case of the Ti felt without coating, the contact resistance is high under low compression force (0.5–2.0 MPa). It is observed that high contact resistance was due to the formation of a less electrically conductive oxide layer on the surface. The formation of the TiO2 surface passivation layer is also confirmed by XPS analysis. The high contact resistances in the case of 0 rpm and 200 rpm are due to no rotating speed and too high a rotating speed during the coating of Ti-PTL, which affects the formation of the gold layer on the porous Ti felt. When the optimized rotating speed was 50 rpm, the lowest contact resistance was 2.0 mΩ cm2 at 1.5 MPa. The low contact resistance was attributed to forming an electrically conductive gold layer on the Ti fibers' outer surface and within the inner volume of the porous Ti felt. The values of interfacial contact resistance (ICR) support the results obtained from Rs of the EIS analysis.
Even after the potentiostatic test, in the case of Ti-PTL treatment under optimized rotating speed (50 rpm), the contact resistance was as low as 9.8 mΩ cm2. However, the uncoated Ti felt, Ti felt coating under no rotation, and Ti felt coating under high rotational speed (200 rpm) showed high contact resistances of 56 mΩ cm2, 38 mΩ cm2, and 27 mΩ cm2 at 1.5 MPa, respectively. It can be concluded that the coating of the porous Ti felt under an optimized rotating speed of 50 rpm did not undergo a significant oxidation of base Ti felt. In addition, ICR results correspond well to the results obtained from the polarization curves of the corrosion study and simulated durability test.
2.5. Performance of gold-coated PTL in a PEMWE cell
The polarization curves were recorded by an in-situ characterization with uncoated Ti felt and gold-coated Ti felt as PTL using a PEMWE cell. In Fig. 8 (c), the Au/Ti felt [R = 50] PTL sample showed better current density-voltage (j-V) performance than uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] PTL samples. The polarization curves verified the high electrical conductivity of Au/Ti felt [R = 50] PTL owing to pinholes-free and cracks-free gold coating on the Ti fibers. The performance of the PEM electrolyzer was improved to 1.742 V at 2.0 A/cm2 with the Au/Ti felt [R = 50] PTL sample, which exceeds values reported in the open literature [3,10,12,[55], [56], [57], [58]]. The high ohmic resistances of the uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] PTL samples increased the cell potential to 2.042 V, 1.965 V, and 1.866 V, respectively, at 2.0 A/cm2. It reveals that the oxidation of titanium fiber on the surface of PTL resulted in the formation of Ti oxides for uncoated Ti felt and samples with pinholes and crack gold coating. With their poor electrical conductivity, the Ti oxides caused the high ohmic resistance observed in the PEM electrolyzer assembled with the uncoated Ti felt, Au/Ti felt [R = 200], and Au/Ti felt [R = 0] PTL samples.
Tafel slope and EIS analysis provide another insight into how the rationale design of the electroplating process for coating PTLs improves PEMWE cells' performance. The iR-free cell voltage curves for PEMWE cells with the uncoated and gold-coated PTLs are shown in Fig. 8 (d). The semi-logarithmic plot presents the role of PTL coating on catalyst layer utilization, specifically for the OER, in the low current density range of 0.01–0.1 A/cm2, where the mass transport resistance is considered negligible. The lower value of the Tafel slope depicts the better OER kinetics at the CL/PTL interface. The gold-coated PTLs exhibit a lower Tafel slope compared to uncoated PTLs, as shown in Fig. 8 (d). The Au/Ti felt [R = 50] showed the lowest value of the Tafel slope. The gold coating on the PTL further reduced the overpotential for electrochemical reactions by enhancing local conductivity at the CL/PTL interface, thereby minimizing losses that would otherwise increase the Tafel slope. This conclusion aligns well with the EIS analysis performed at 1A/cm2, shown in the Nyquist plot (Fig. 8 (e)). The sample with a uniform gold coating (Au/Ti felt [R = 50]) showed the lowest value of mass transfer resistance due to the efficient removal of oxygen bubbles at a current density of 1 A/cm2. Therefore, the polarization curve of the Au/Ti felt [R = 50] PTL showed a slight deviation in the Tafel slope at high current densities compared to the deviations in the Tafel slope of the uncoated Ti felt, Au/Ti felt [R = 0]. Au/Ti felt [R = 200] PTLs.
In Fig. 8(e), the EIS spectra of gold-coated PTLs consisted of two semicircles ranging from 40 kHz to 0.1 Hz. The first semicircle was observed at a high frequency from 40 kHz to 100 Hz, and the second at a lower frequency from 100 Hz to 0.1 Hz. The leftmost x-intercept of the EIS curves represents the ohmic resistance of the PEMWE cell. In contrast, the rightmost x-intercepts indicate the total resistances, including the ohmic resistance (RΩ), charge-transfer resistance (Rct), and mass transfer resistance (Rmt). The values of RΩ, Rct, and Rmt are presented in Table 2. The semicircles corresponding to mass transfer resistance are very small at 1 A/cm2, indicating that mass transport loss has limited effects on the performance of the PEMWE cell. The values of Rct were approximately 34 mΩ cm2, 45 mΩ cm2, and 70 mΩ cm2 for Au/Ti felt [R = 50], Au/Ti felt [R = 0], and Au/Ti felt [R = 200], respectively.
Table-2. The equivalent circuits of EIS spectra fitted by the Z-view software.
| PTL sample | RΩ (mΩ cm2) | Rct (mΩ cm2) | Rmt (mΩ cm2) | Rp (mΩ cm2) |
| Uncoated Ti felt | 270 | 66 | 18 | 20 |
| Au/Ti felt [R = 0] | 150 | 45 | 8 | – |
| Au/Ti felt [R = 50] | 120 | 34 | 2 | – |
| Au/Ti felt [R = 200] | 220 | 70 | 12 | – |
In general, the diameter of the semicircle, which is associated with charge transfer resistance, reflects the OER kinetics [3,12,[59], [60], [61]]. The Au/Ti felt [R = 50] sample showed the lowest charge transfer resistance, indicating improved OER kinetics compared to other counterpart PTLs. The EIS spectrum of the uncoated Ti felt differs significantly, exhibiting three distinct semicircles. One additional semicircle at a high frequency from 40 kHz to 2 kHz is attributed to an electrical contact resistance (Rp) at the interface of the catalyst layer and PTL [12,62]. Since the high-frequency semicircle was observed only for the uncoated Ti felt, it can clearly be attributed to the presence of a passivated oxide layer that impeded the performance of the PEMWE cell.
The uniform gold coating of PTL under substrate rotation of 50 rpm made a strong contact at the CL/PTL interface, which is not only improved kinetics but also reduced the value of high-frequency resistance (HFR). Typically, HFR is the summation of electrical contact resistances and membrane resistance [3,13]. In this study, membrane resistance is similar for all four PTLs because the technical specification of CCM is the same. The protective layer of gold has modified the inner and outer structure of PTL, increasing the electrically conductive surface area in contact with the catalyst layer while avoiding the deformation of the membrane into the PTL with the compression. In Fig. 8 (f), the HFR value vs. current density curve was obtained by EIS technique. At low current densities of 0–0.2 A/cm2, the PEMWE cell showed a sharp drop in the HFR value. At very low current densities (0–0.2 A/cm2), the rate of H⁺ formation is low, resulting in an insignificant osmotic water drag through the membrane. Although the PEM was not actively hydrating at low current densities, the water content still played a role in determining proton conductivity [63,64]. The HFR values of Au/Ti felt [R = 50] PTL were lowest and remained constant at current densities of 0.2, 0.4, 0.6, 0.8, and 1.0 A/cm2. The uniformly gold-coated PTL facilitated the rapid detachment of oxygen bubbles, which may further result in a low HFR at high current density [65,66]. The HFR values of uncoated Ti felt Au/Ti felt [R = 0], and Au/Ti felt [R = 200] samples increase at high current densities. This is likely due to the combined effect of high electrical resistance at the CL/PTL interface and oxygen bubble accumulation near the catalyst surface at high current densities, which caused a local dehydration of the membrane.
It can be concluded that the engineered gold coating strategy has significantly improved electrical contact at the CL/PTL interface. This method holds promise for enhancing PEMWE cell performance due to its advanced technology and potential for easy commercialization. In addition, to separate the contributions of individual phenomena, we developed a PTL testing device to simulate the PEM electrolyzer condition without using CCM and metallic bipolar plates. The fabrication of non-metallic BPs using 3D printing offered a simulated flow field structure at the PTL/BPs interface. This approach paves the way for independent observation of the impact on cell voltage caused by the degradation of the PTL.
3. Conclusions
In this study, the degradation of Ti-PTL was mitigated using a three-step process: electrochemical etching, Au electrostriking, and Au electroplating. Electrochemical etching removed surface oxides, Au electrostriking created an Au underlayer by passivating oxides, and electroplating added a protective top layer of gold. Using a rotation speed of 50 rpm during the coating process, the Ti-PTL showed better corrosion resistance than no rotation or a high-speed rotation of 200 rpm. 3D printing was used to make non-metallic bipolar plates with a simulated flow field structure at the interface of the Ti-PTL and the bipolar plate. The simulated conditions, like the solution temperature (80 °C), flow rate (50 mL min−1), solution concentration (0.5 M H2SO4), and cell potential (2.0 V), were set to mimic a conventional PEM water electrolyzer. The Au/Ti felt [R = 50] showed better durability than both Au/Ti felt [R = 200] and Au/Ti felt [R = 0], with fewer uncoated areas prone to corrosion in harsh acidic conditions. Using Au/Ti felt [R = 50], a single-cell performance of 1.742 V at 2.0 A/cm2 was achieved at 80 °C under ambient pressure, with a precious metal loading of 1 mg/cm2 in the anodic catalyst layer. The engineered gold electroplating process improved the coating on the inner and outer surfaces of the Ti-PTL, increasing the conductive area in contact with the catalyst. The gold-coated porous Ti felt showed excellent stability, making it a promising option for improving PEM water electrolyzer performance and scaling up for larger production.
Written by Aditya Singh, Biswajit Samir De, Sukhvant Singh, Sai Praneeth Thota, Mohmmad Khalid, and Samaneh Shahgaldi; from the Hydrogen Research Institute, Universite Du Quebec a Trois-Rivieres, Trois-Rivieres, Quebec, G8Z 4M3, Canada
CRediT authorship contribution statement: Aditya Singh: Writing – original draft, Visualization, Validation, Methodology, Investigation, Data curation, Conceptualization. Biswajit Samir De: Methodology, Investigation, Formal analysis. Sukhvant Singh: Methodology, Investigation, Formal analysis, Data curation. Sai Praneeth Thota: Visualization, Formal analysis. Mohmmad Khalid: Formal analysis. Samaneh Shahgaldi: Writing – review & editing, Supervision, Resources, Funding acquisition, Formal analysis.
Declaration of interest statement: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments: This project received financial support from Mitacs Accelerate (Application Ref.: IT29926) with Intlvac Canada, Natural Sciences and Engineering Research Council of Canada (NSERC), the Canada Research Chair program (CRC-2019-00354) and the Discovery Grant (CRSNG-DGECR-2022-00058).
References
[1] Yuan XZ, Shaigan N, Song C, Aujla M, Neburchilov V, Kwan JTH, Wilkinson DP, Bazylak A, Fatih K. The porous transport layer in proton exchange membrane water electrolysis: perspectives on a complex component. Sustain Energy Fuels 2022;6: 1824–53.
[2] Wilberforce T, Ijaodola O, Ogungbemi E, Khatib FN, Leslie T, El-Hassan Z, Thomposon J, Olabi AG. Technical evaluation of proton exchange membrane (PEM) fuel cell performance – a review of the effects of bipolar plates coating. Renew Sustain Energy Rev 2019;113:109286.
[3] Liu C, Wippermann K, Rasinski M, Suo Y, Shviro M, Carmo M, Lehnert W. Constructing a multifunctional interface between membrane and porous transport layer for water electrolyzers. ACS Appl Mater Interfaces 2021;13:16182–96.
[4] Yasin MC, Johar M, Gupta A, Shahgaldi S. A comprehensive review of the material innovations and corrosion mitigation strategies for PEMWE bipolar plates. Int J Hydrogen Energy 2024;88:726–47.
[5] Prestat M. Corrosion of structural components of proton exchange membrane water electrolyzer anodes: a review. J Power Sources 2023;556:232469.
[6] Fan HQ, Wu YM, Su S, Shi DD, Wang XZ, Behnamian Y, Zhang JY, Li Q. Solution acidity and temperature induced anodic dissolution and degradation of through- plane electrical conductivity of Au/TiN coated metal bipolar plates used in PEMFC. Energy 2022;254:124453.
[7] Stiber S, Sata N, Morawietz T, Ansar SA, Jahnke T, Lee JK, Bazylak A, Fallisch A, Gago AS, Friedrich KA. A high-performance, durable and low-cost proton exchange membrane electrolyser with stainless steel components. Energy Environ Sci 2022; 15:109–22.
[8] Feng Q, Yuan XZ, Liu G, Wei B, Zhang Z, Li H, Wang H. A review of proton exchange membrane water electrolysis on degradation mechanisms and mitigation strategies. J Power Sources 2017;366:33–55.
[9] Tan A, Zhang Y, Shi X, Ju C, Liu P, Yang T, Liu J. The poisoning effects of Ti-ion from porous transport layers on the membrane electrode assembly of proton exchange membrane water electrolyzers. Chem. Eng. J. 2023;471:144624.
[10] Liu C, Carmo M, Bender G, Everwand A, Lickert T, Young JL, Smolinka T, Stolten D, Lehnert W. Performance enhancement of PEM electrolyzers through iridium-coated titanium porous transport layers. Electrochem Commun 2018;97: 96–9.
[11] Doan TL, Lee HE, Shah SSH, Kim MJ, Kim CH, Cho HS, Kim T. A review of the porous transport layer in polymer electrolyte membrane water electrolysis. Int J Energy Res 2021;45:14207–20.
[12] Rakousky C, Keeley GP, Wippermann K, Carmo M, Stolten D. The stability challenge on the pathway to high-current-density polymer electrolyte membrane water electrolyzers. Electrochim Acta 2018;278:324–31.
[13] Liu C, Shviro M, Gago AS, Zaccarine SF, Bender G, Gazdzicki P, Morawietz T, Biswas I, Rasinski M, Everwand A, Schierholz R, Pfeilsticker J, Müller M, Lopes PP, Eichel RA, Pivovar B, Pylypenko S, Friedrich KA, Lehnert W, Carmo M. Exploring the interface of skin-layered titanium fibers for electrochemical water splitting. Adv Energy Mater 2021;11.
[14] Gupta A, Chellehbari YM, Shahgaldi S. Achieving high performance and durability with ultra-low precious metal nanolayer on porous transport layer for PEMWE application. J Power Sources 2025;630:236088.
[15] Kang Z, Mo J, Yang G, Li Y, Talley DA, Retterer ST, Cullen DA, Toops TJ, Brady MP, Bender G, Pivovar BS, Green JB, Zhang FY. Thin film surface modifications of thin/ tunable liquid/gas diffusion layers for high-efficiency proton exchange membrane electrolyzer cells. Appl Energy 2017;206:983–90.
[16] Wang Q, Zhou Z, Ye K, Hu M, Hu X, Wang S, Hu C. The effect of pretreatment and surface modification of porous transport layer (PTL) on the performance of proton exchange membrane water electrolyzer. Int J Hydrogen Energy 2024;53:163–72.
[17] Srour T, Kumar K, Martin V, Dubau L, Maillard F, Gilles B, Dillet J, Didierjean S, Amoury B, Le TD, Maranzana G. On the contact resistance between the anode and the porous transport layer in a proton exchange membrane water electrolyzer. Int J Hydrogen Energy 2024;58:351–61.
[18] Liu H, Ma D, Li Y, You L, Leng Y. Evolution of the shadow effect with film thickness and substrate conductivity on a hemispherical workpiece during magnetron sputtering. Metals 2023;13.
[19] Yang G, Yu S, Mo J, Kang Z, Dohrmann Y, List FA, Green JB, Babu SS, Zhang FY. Bipolar plate development with additive manufacturing and protective coating for durable and high-efficiency hydrogen production. J Power Sources 2018;396: 590–8. 1039 A. Singh et al. [20] Giurlani W, Zangari G, Gambinossi F, Passaponti M, Salvietti E, Di Benedetto F, Caporali S, Innocenti M. Electroplating for decorative applications: recent trends in research and development. Coatings 2018;8:1–25.
[21] Choi JH, Eun Kang H, Kim DJ, Soo Yoon Y. A comprehensive review of stainless- steel bipolar plate coatings and their role in mitigating corrosion in aggressive proton-exchange membrane fuel cells environments. Chem. Eng. J. 2024;493: 152662.
[22] Cherevko S, Topalov AA, Zeradjanin AR, Katsounaros I, Mayrhofer KJJJ, Keeley GP, Mayrhofer KJJJ. A comparative study on gold and platinum dissolution in acidic and alkaline media. J Electrochem Soc 2014;3:H822–30.
[23] Cherevko S, Topalov AA, Zeradjanin AR, Katsounaros I, Mayrhofer KJJ. Gold dissolution: towards understanding of noble metal corrosion. RSC Adv 2013;3: 16516–27.
[24] Liu Y, Huang S, Wang D, Zhang H, Shan D, Peng S, Shen G, Wang L, Wang X. Modifying Ti-based gas diffusion layer passivation for polymer electrolyte membrane water electrolysis via electrochemical nitridation. ACS Appl Mater Interfaces 2022;14:15728–35.
[25] Gago AS, Ansar SA, Saruhan B, Schulz U, Lettenmeier P, Ca˜ nas NA, Gazdzicki P, Morawietz T, Hiesgen R, Arnold J, Friedrich KA. Protective coatings on stainless steel bipolar plates for proton exchange membrane (PEM) electrolysers. J Power Sources 2016;307:815–25.
[26] Samir De B, Singh A, Ji Dixit R, Khare N, Elias A, Basu S. Hydrogen generation in additively manufactured membraneless microfluidic electrolysis cell: performance evaluation and accelerated stress testing. Chem. Eng. J. 2023;452:139433.
[27] Yang G, Yu S, Kang Z, Dohrmann Y, Bender G, Pivovar BS, Green JB, Retterer ST, Cullen DA, Zhang FY, Ambrosi A, Pumera M. A novel PEMEC with 3D printed non- conductive bipolar plate for low-cost hydrogen production from water electrolysis. ACS Sustainable Chem Eng 2018;6:16968–75.
[28] Ambrosi A, Pumera M. Multimaterial 3D-printed water electrolyzer with earth- abundant electrodeposited catalysts. ACS Sustainable Chem Eng 2018;6:16968–75.
[29] Wen J, Allain N, Fleury E. Determination of orientation relationships between FCC- hydride and HCP-titanium and their correlation with hydrides distribution. J Alloys Compd 2020;817:153297.
[30] Wu TI, Wu JK. Effects of electrolytic hydrogenating parameters on structure and composition of surface hydrides of CP-Ti and Ti-6Al-4V alloy. Mater Chem Phys 2002;74:5–12.
[31] Wen J, Allain N, Fleury E, Wu TI, Wu JK, Bautkinova T, Utsch N, Bystron T, Lhotka M, Kohoutkova M, Shviro M, Bouzek K. Introducing titanium hydride on porous transport layer for more energy efficient water electrolysis with proton exchange membrane. J Alloys Compd 2020;565:232913.
[32] Skvorˇ cinskien˙ e R, Urbonaviˇ cius M, Vorotinskien˙ e L, Baˇ sinskas M, Zakarauskas K, Maziukien˙ e M, Stri¯ ugas N, Buidovas P, Milˇ cius D. Varnagiris, Application of TiH2 dehydrogenation for vapour layer formation under boiling crisis conditions. Appl Therm Eng 2024;247.
[33] Skvorˇ cinskien˙ e R, Urbonaviˇ cius M, Vorotinskien˙ e L, Baˇ sinskas M, Zakarauskas K, Maziukien˙ e M, Stri¯ ugas N, Buidovas P, Milˇ cius D, Varnagiris, Frank MJ, Walter MS, Lyngstadaas SP, Wintermantel E, Haugen HJ. Hydrogen content in titanium and a titanium-zirconium alloy after acid etching. Mater Sci Eng C 2013;247:1282–8.
[34] Lettenmeier P, Wang R, Abouatallah R, Saruhan B, Freitag O, Gazdzicki P, Morawietz T, Hiesgen R, Gago AS, Friedrich KA. Low-cost and durable bipolar plates for proton exchange membrane electrolyzers. Sci Rep 2017;7:1–12.
[35] Yuan H, Li J, Tang Z, Wang Y, Wu T, Huang M, Zhao L, Zhao Z, Liu H, Xu C, Liu X, Zhou W. Enhanced interfacial stability of Pt/TiO2/Ti via Pt-O bonding for efficient acidic electrolyzer. Chem. Eng. J. 2024;492:152339.
[36] Aßmann P, Gago AS, Gazdzicki P, Friedrich KA, Wark M. Toward developing accelerated stress tests for proton exchange membrane electrolyzers. Curr Opin Electrochem 2020;21:225–33.
[37] Li K, Yu S, Li D, Ding L, Wang W, Xie Z, Park EJ, Fujimoto C, Cullen DA, Kim YS, Zhang FY. Engineered thin diffusion layers for anion-exchange membrane electrolyzer cells with outstanding performance. ACS Appl Mater Interfaces 2021; 13:50957–64.
[38] Xu J, Huang HJ, Li Z, Xu S, Tao H, Munroe P, Xie ZH. Corrosion behavior of a ZrCN coated Ti alloy with potential application as a bipolar plate for proton exchange membrane fuel cell. J Alloys Compd 2016;663:718–30.
[39] Conde JJ, Ferreira-Aparicio P, Chaparro AM. Anti-corrosion coating for metal surfaces based on superhydrophobic electrosprayed carbon layers. Appl Mater Today 2018;13:100–6.
[40] Wang SH, Peng J, Lui WB, Zhang JS, Navinˇ sek B, Panjan P, Miloˇ sev I. PVD coatings as an environmentally clean alternative to electroplating and electroless processes. Surf Coating Technol 2006;162:476–87.
[41] Wang SH, Peng J, Lui WB, Zhang JS. Performance of the gold-plated titanium bipolar plates for the light weight PEM fuel cells. J Power Sources 2006;162: 486–91.
[42] Kellenberger A, Vaszilcsin N, Duca D, Dan ML, Duteanu N, Stiber S, Morawietz T, Biswas I, Ansar SA, Gazdzicki P, Wirkert FJ, Roth J, Rost U, Brodmann M, Gago AS, International Journal of Hydrogen Energy 106 (2025) 1029–1040 Friedrich KA. Towards replacing titanium with copper in the bipolar plates for proton exchange membrane water electrolysis. Materials 2022;15.
[43] Doan TL, Lee HE, Kim MJ, Cho WC, Cho HS, Kim T. Influence of IrO2/TiO2 coated titanium porous transport layer on the performance of PEM water electrolysis. J Power Sources 2022;533:231370.
[44] Tan Q, Wang Y. Preparation and performances of modified Ti4O7 doped polypyrrole coating for metallic bipolar plates. Corros Sci 2021;190:109703.
[45] Wakayama H, Yamazaki K. Low-cost bipolar plates of Ti4O7-coated Ti for water electrolysis with polymer electrolyte membranes. ACS Omega 2021;6:4161–6.
[46] S´ anchez Batalla B, Laube A, Hofer A, Struckmann T, Bachmann J, Weidlich C. Degradation studies of proton exchange membrane water electrolysis cells with low platinum group metals – catalyst coating achieved by atomic layer deposition. Int J Hydrogen Energy 2022;47:39719–30.
[47] Van Pham C, Escalera-L´ opez D, Mayrhofer K, Cherevko S, Thiele S. Essentials of high performance water electrolyzers – from catalyst layer materials to electrode engineering. Adv Energy Mater 2021;11.
[48] Gao X, Xu G, Zhao Y, Li S, Shi F, Chen Y. Self-assembly of amine-functionalized gold nanoparticles on phosphonate-functionalized graphene nanosheets: a highly active catalyst for the reduction of 4-nitrophenol. RSC Adv 2015;5:88045–51.
[49] Bilovol V, Ferrari S, Derewnicka D, Saccone FD. XANES and XPS study of electronic structure of Ti-enriched Nd-Fe-B ribbons. Mater Chem Phys 2014;146:269–76.
[50] Xie W, Li R, Xu Q. Enhanced photocatalytic activity of Se-doped TiO2 under visible light irradiation. Sci Rep 2018;8:1–10.
[51] Tiwari M, Singh A, Thakur D, Pattanayek SK. Graphitic carbon nitride-based concoction for detection of melamine and R6G using surface-enhanced Raman scattering. Carbon N. Y. 2022;197:311–23.
[52] Zhang X, Shi H, Xu B. Catalysis by gold: isolated surface Au 3+ions are active sites for selective hydrogenation of 1,3-butadiene over Au/ZrO 2 catalysts. Angew. Chemie 2005;117:7294–7.
[53] Gougis M, Pereira A, Ma D, Mohamedi M. Simultaneous deposition of cerium oxide and gold nanostructures-characterization and analytical properties toward glucose electro-oxidation and sensing. RSC Adv 2014;4:39955–61.
[54] Cherevko S, Topalov AA, Katsounaros I, Mayrhofer KJJ. Electrochemical dissolution of gold in acidic medium. Electrochem Commun 2013;28:44–6.
[55] Fan Z, Yu H, Jiang G, Yao D, Sun S, Chi J, Qin B, Shao Z. Low precious metal loading porous transport layer coating and anode catalyst layer for proton exchange membrane water electrolysis. Int J Hydrogen Energy 2022;47:18963–71.
[56] Grigoriev SA, Millet P, Volobuev SA, Fateev VN. Optimization of porous current collectors for PEM water electrolysers. Int J Hydrogen Energy 2009;34:4968–73.
[57] Yasutake M, Noda Z, Matsuda J, Lyth SM, Nishihara M, Ito K, Hayashi A, Sasaki K. Ru-core Ir-shell electrocatalysts deposited on a surface-modified Ti-based porous transport layer for polymer electrolyte membrane water electrolysis. Int J Hydrogen Energy 2024;49:169–83.
[58] Majasan JO, Iacoviello F, Shearing PR, Brett DJL. Effect of microstructure of porous transport layer on performance in polymer electrolyte membrane water electrolyser. Energy Proc 2018;151:111–9.
[59] Singh A, Karmakar S, Basu S. Role of sputtered WO3 underlayer and NiFeCr-LDH co-catalyst in WO3–BiVO4 heterojunction for enhanced photoelectrochemical water oxidation. Int J Hydrogen Energy 2021;46:39868–81.
[60] Singh A, De BS, Karmakar S, Basu S. Large-area WO3/BiVO4-CoPi photoanode for efficient photoelectrochemical water splitting: role of patterned metal microgrid and electrolyte flow. ACS Appl Energy Mater 2023;6:4642–56.
[61] Sarma SK, Singh A, Mohan R, Shukla A. Computational fluid dynamics simulation of bubble hydrodynamics in water splitting: effect of electrolyte inflow velocity and electrode morphology on cell performance. Int J Hydrogen Energy 2023;48: 17769–82.
[62] Bernt M, Schramm C, Schr¨ oter J, Gebauer C, Byrknes J, Eickes C, Gasteiger HA. Effect of the IrO x conductivity on the anode electrode/porous transport layer interfacial resistance in PEM water electrolyzers. J Electrochem Soc 2021;168: 084513.
[63] Kosakian A, Urbina LP, Heaman A, Secanell M. Understanding single-phase water- management signatures in fuel-cell impedance spectra: a numerical study. Electrochim Acta 2020;350:136204.
[64] Immerz C, Bensmann B, Trinke P, Suermann M, Hanke-Rauschenbach R. Local current density and electrochemical impedance measurements within 50 cm single-channel PEM electrolysis cell. J Electrochem Soc 2018;165:F1292–9.
[65] Yuan S, Zhao C, Cai X, An L, Shen S, Yan X, Zhang J. Bubble evolution and transport in PEM water electrolysis: mechanism, impact, and management. Prog Energy Combust Sci 2023;96.
[66] Yuan S, Zhao C, Cai X, An L, Shen S, Yan X, Zhang J, Hu Y, Yang Y, Zeng Z, Zhou T, Yang F, Sun W, He L. Preliminary investigation of factors influencing the bubbles detachment of iridium oxide in oxygen evolution reaction. Int J Hydrogen Energy 2024;61:754–61.



































