If the steel substrate contains lead, be sure to verify that the acid pickle contains a sufficient amount of bilfuoride to adequately remove the lead from the surface, or poor adhesion may result.
 Frank AltmayerAdhesion problems on ferrous parts can result from absorption of hydrogen gas during plating. All steel absorbs hydrogen gas during (cathodic) electrocleaning, acid pickling and plating. Adhesion failures consisting of tiny blisters are indica- tive of hydrogen gas leaving the steel during exposure of the part at elevated temperature. Possible cures include use of inhibitors in the acid, plating in more efficient solutions, and a slow ramp bake at elevated temperature after plating.
Frank AltmayerAdhesion problems on ferrous parts can result from absorption of hydrogen gas during plating. All steel absorbs hydrogen gas during (cathodic) electrocleaning, acid pickling and plating. Adhesion failures consisting of tiny blisters are indica- tive of hydrogen gas leaving the steel during exposure of the part at elevated temperature. Possible cures include use of inhibitors in the acid, plating in more efficient solutions, and a slow ramp bake at elevated temperature after plating.
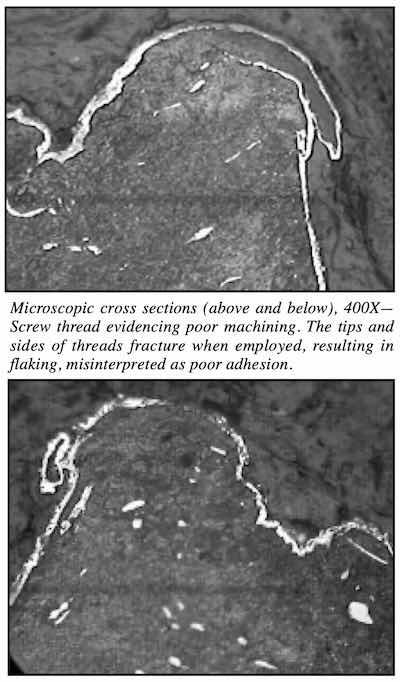
Adhesion failures can also be “misdiagnosed” machining problems. One recently experienced example is flaking of machined steel threads related to the rough threads them- selves. The surface of the threads was so rough and deformed that steel, along with the plating, fractured off when the threads were employed. This was erroneously considered to be a plating failure. The first two photos illustrate the type of rough-machined surface a threaded part may have.
Because a majority of steel parts are zinc plated, adhesion failures of zinc-plated steel parts are frequently encountered. Zinc can be plated from a variety of chemistries, but the three most common offer varying degrees of ductility. The non-cyanide zinc solutions tend to yield brittle deposits when the plating thickness approaches or exceeds 10 microns (0.0004 in.). The deposit may be brittle, resulting in zinc flaking off itself. Lowering the brightener content (if possible), or using a cyanide process, yields a more ductile deposit.
 Poor Adhesion: Aluminum Substrates Adhesion failures on aluminum parts are most often related to the part itself, if it is a die casting, or the zincate immersion deposit applied ahead of plating.
Poor Adhesion: Aluminum Substrates Adhesion failures on aluminum parts are most often related to the part itself, if it is a die casting, or the zincate immersion deposit applied ahead of plating.
Aluminum Die Castings: A cross section of an aluminum casting should not show a significant number of surface pores, especially large ones. The plater can do little about these, because they are a problem related to the manufacture of the casting. The die temperature, melt temperature, gating and injection rates all play a role in the degree of surface porosity in the structure of the die cast part.
Zincate Dip: Aluminum parts must be processed to remove and replace an oxide film that forms “instantly” over cleaned aluminum. While proprietary systems are available, many platers utilize a zincate solution to obtain an immer- sion zinc deposit over the aluminum prior to a copper strike to obtain an initial adherent deposit over the aluminum.
An adhesion problem on an aluminum substrate may be related to this zincate operation. Assuming the zincate solution is of the correct concentration, things to look for include:
Contamination by Other Heavy Metals: In one instance, we discovered that whenever the cadmium content of the zincate reached 9 ppm, we had to replace it, or face higher rejects caused by blistering of nickel- cadmium-plated aluminum connector shells, when exposed at elevated temperature. Most heavy metals more noble than zinc must be kept out of the zincating solution. An exception is iron, which often is added to modify the crystal structure of the zinc deposit.
Thick-Porous Zinc Deposits: A zincate dip is normally used twice. The parts are zincated, stripped in nitric acid and re-zincated. Experience has indicated that a two-step zincate yields a denser, thinner layer of zinc. One can also obtain a porous, less adherent zinc film by operating the zincate at too high a temperature, leaving the parts in the solution too long, or having an improper zinc-to- sodium hydroxide ratio. Our experi- ence has been that a single dip in a “modified” zincate formulation can yield zinc films that are superior to a “conventional” zincate, even when double-dipped. The modified zincate can reduce pollution loading as well, because it is almost half the con- centration of a conventional solution.
 Other Possible Causes: Of Poor Adhesion on Aluminum The zincated aluminum most often is plated in a cyanide copper strike solution that is specially formulated to avoid immersion deposits over the zinc film. The strike solution should not be too high in pH, because zinc dissolves at elevated pH values. The copper and free cyanide content play a major role in avoiding immersion deposits as well. Watch out for high copper concentrations, low free cyanide, high temperatures and hexavalent chromium contamination in the copper strike as well.
Other Possible Causes: Of Poor Adhesion on Aluminum The zincated aluminum most often is plated in a cyanide copper strike solution that is specially formulated to avoid immersion deposits over the zinc film. The strike solution should not be too high in pH, because zinc dissolves at elevated pH values. The copper and free cyanide content play a major role in avoiding immersion deposits as well. Watch out for high copper concentrations, low free cyanide, high temperatures and hexavalent chromium contamination in the copper strike as well.
Aluminum alloys containing silicon require acid pickling in fluoride-containing acids to remove surface silicates, which inhibit adhesion. The acid typically used in electroplating aluminum components is based on nitric acid, 25-50 percent volume, usually containing ammonium bifluoride at 1.5-8 oz/gal (10-60 g/L).
It has been reported that etching aluminum parts as follows signifi- cantly enhances the adhesion of plated deposits on aluminum alloys.
- Phosphoric Acid (H3PO4): 10% vol.
- Sulfuric Acid (H2SO4): 5% vol.
- Temperature, C: 65
- Time, min: 2
or
- Nitric acid (HNO3): 2.5% vol.
- Sulfuric acid (H2SO4): 1% vol.
- Ammonium bifluoride (NH4HF2): 3% wt. Temperature, C: 30
- Time, min: 2
Etching the surface to be plated creates “anchor points” for plated metal to hold onto, thereby improving the adhesion. It also increases the surface area that a de-cohesive force must work against, resulting in a lower force per unit area than would be necessary for the deposit to not adhere in the absence of an etch.
Poor Adhesion of Zinc Substrates: Parts made of zinc are typically made using the die casting process described above, and are subject to the same surface porosity problems. Similarly, they can suffer adhesion failures if the cyanide copper strike is not well controlled, as described above. Because zinc is a very active metal, be sure that the cleaners are not too high in alkalinity, and the acid isn’t too strong, because both will tend to attack and etch the zinc, possibly leaving residues that will yield adhesion problems.
Poor Adhesion of Copper Alloys: A wide variety of copper alloys are plated, including brass, bronze, copper- beryllium and copper tellurium alloys. The beryllium alloy is found in copper parts that are heat treated, or require high wear resistance. Heat-treated parts evidence a thick oxide film that must be completely pickled off. Tellurium is added to copper to enhance the machinability. The preparatory cycle should merit close attention, because close control over the chemical composition is more important for this alloy.
Process-related Adhesion Problems for Copper and Copper Alloys: Poor adhesion may be caused by excessive reverse electrocleaning on most all copper alloys. Reverse electrocleaning has a tendency to produce copper oxides, which some acid pickles cannot easily remove. Aluminum and silicon bronze alloys are typically pickled in nitric- bifluoride to remove aluminum and silicon contamination from the surface after cleaning. Concentrate on the pickle first. The bifluoride concentration in the nitric acid is important to control.
Poor Adhesion of Stainless Steel: Poor adhesion to stainless steel parts typically is related to the strike solution. The strike must be of low efficiency, so as to reduce surface oxides, while a thin film of nickel is deposited. Strike formulations based on nickel chloride-hydrochloric (Wood’s) and nickel sulfamate- sulfamic acid are common.
Poor Adhesion of Cast Iron: Cast iron contains graphite, which leaves heavy smut over the surface that is difficult to remove (without creating more smut) if the acid pickle is too strong or the parts are pickled for too long a time. The presence of these stringers on the surface can result in shearing off of particles (see above photos) in wear applications, which can be misinterpreted as poor adhesion.
Frank Altmayer is a Master Surface Finisher, an AESF Fellow, and the technical education director of the AESF Foundation and NASF. He owned Scientific Control Laboratories from 1986 to 2007 and has over 50 years of experience in metal finishing. He received the AESF Past Presidents Award, NAMF Award of Special Recognition, AESF Leadership Award, AESF Fellowship Award, Chicago Branch AESF Geldzahler Service Award, and NASF Award of Special Recognition.



































